Lewis Dot Structure For Sf2
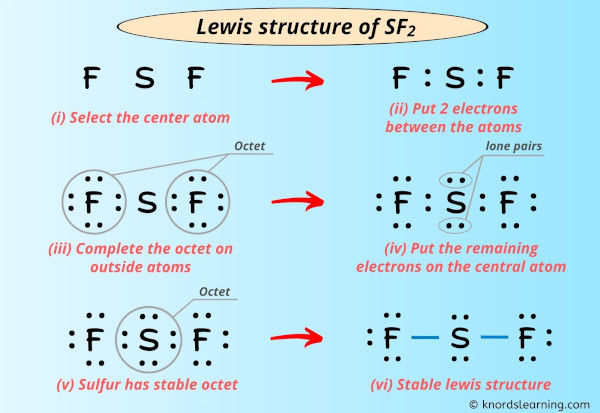
I'm super excited to teach you the lewis structure of SF2 in just half-dozen simple steps.
Infact, I've too given the step-by-step images for drawing the lewis dot construction of SF2 molecule.
So, if you are set to go with these 6 simple steps, then permit's dive right into it!
Lewis structure of SF2 contains two unmarried bonds betwixt the Sulfur (S) cantlet and each Fluorine (F) atom. The Sulfur atom (S) is at the heart and information technology is surrounded by 2 Fluorine atoms (F). The Sulfur cantlet has 2 solitary pairs and both the Fluorine atoms accept three lone pairs.
Let'south depict and empathise this lewis dot structure stride past step.
(Note: Take a pen and newspaper with you and effort to draw this lewis structure along with me. I am sure you will definitely learn how to draw lewis structure of SF2).
6 Steps to Draw the Lewis Construction of SF2
Step #i: Calculate the total number of valence electrons
Here, the given molecule is SF2 (sulfur difluoride). In lodge to draw the lewis structure of SF2, first of all you have to find the total number of valence electrons present in the SF2 molecule.
(Valence electrons are the number of electrons present in the outermost shell of an atom).
And then, let'south summate this first.
Calculation of valence electrons in SF2
- For Sulfur:
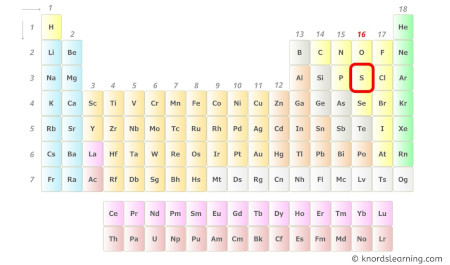
Sulfur is a group sixteen chemical element on the periodic table.
Hence, the valence electrons present in sulfur is 6 (see below image).

- For Fluorine:
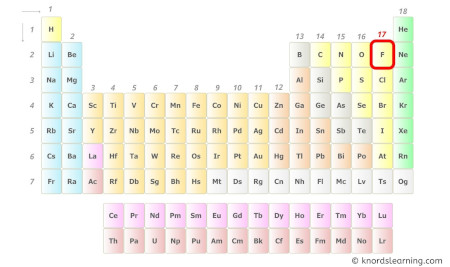
Fluorine is a group 17 chemical element on the periodic table.
Hence, the valence electrons present in fluorine is vii (see below image).

Hence in a SF2 molecule,
Valence electrons given by Sulfur (S) atom = half-dozen
Valence electrons given past each Fluorine (F) cantlet = 7
So, total number of Valence electrons in SF2 molecule = 6 + 7(2) = 20
Step #ii: Select the centre atom
While selecting the atom, always put the least electronegative atom at the centre.
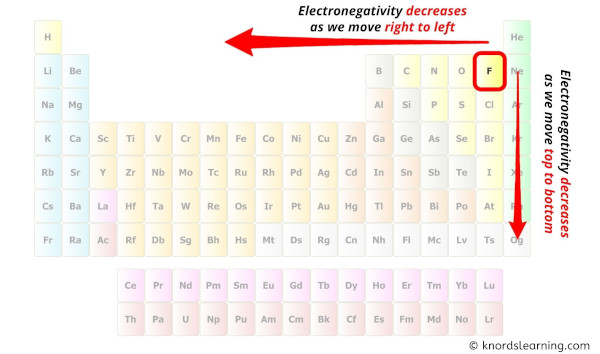
(Remember: Fluorine is the most electronegative element on the periodic table and the electronegativity decreases as nosotros move right to left in the periodic tabular array equally well as height to bottom in the periodic tabular array).
Here in the SF2 molecule, if we compare the sulfur atom (S) and fluorine atom (F), then the sulfur is less electronegative than fluorine.
So, sulfur should be placed in the center and the remaining ii fluorine atoms will surround it.
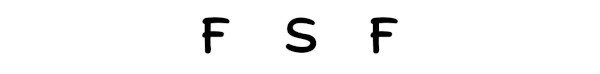
Step #3: Put two electrons between the atoms to represent a chemical bail
At present in the higher up sketch of SF2 molecule, put the two electrons (i.e electron pair) between each sulfur atom and fluorine atom to stand for a chemical bail between them.

These pairs of electrons present between the Sulfur (S) and Fluorine (F) atoms form a chemical bond, which bonds the sulfur and fluorine atoms with each other in a SF2 molecule.
Pace #4: Complete the octet (or duplet) on outside atoms. If the valence electrons are left, then put the valence electrons pair on the central atom
Don't worry, I'll explain!
In the Lewis construction of SF2, the outer atoms are fluorine atoms.
So now, you have to complete the octet on these fluorine atoms (because fluorine requires 8 electrons to accept a complete outer vanquish).
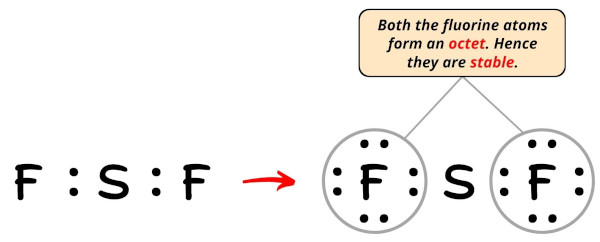
Now, you can see in the higher up image that all the fluorine atoms form an octet.
Too, only 16 valence electrons of SF2 molecule are used in the above structure.
Merely there are total 20 valence electrons in SF2 molecule (equally calculated in footstep #one).
Then the number of electrons left to be kept on the central atom = 20 – sixteen = 4.
Then permit's go along these four electrons (i.eastward 2 electron pairs) on the key atom.
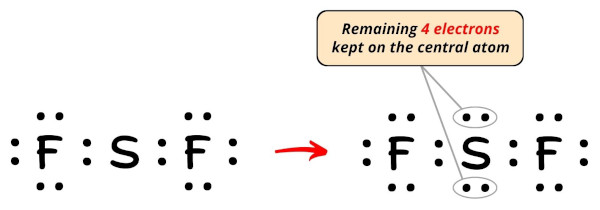
Now, let'southward move to the next step.
Pace #five: Check whether the central cantlet has octet or not. If it does non have an octet, so motility the electron pair from the outer cantlet to form a double bond or triple bond
In this stride, we take to check whether the central atom (i.e sulfur) has an octet or not.
In simple words, we accept to check whether the central Sulfur (S) atom is having 8 electrons or not.
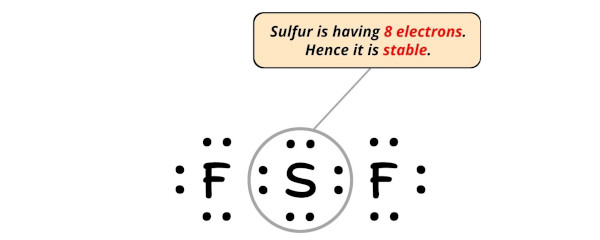
As you tin see from the to a higher place prototype, the primal atom (i.eastward sulfur), has 8 electrons. So it fulfills the octet rule and the sulfur cantlet is stable.
Step #6: Final step – Check the stability of lewis construction by calculating the formal accuse on each atom
At present, you accept come up to the concluding step and hither you lot have to cheque the formal charge on sulfur atom (S) too equally each fluorine cantlet (F).
For that, you need to think the formula of formal charge;
Formal charge = Valence electrons – Nonbonding electrons – (Bonding electrons)/two
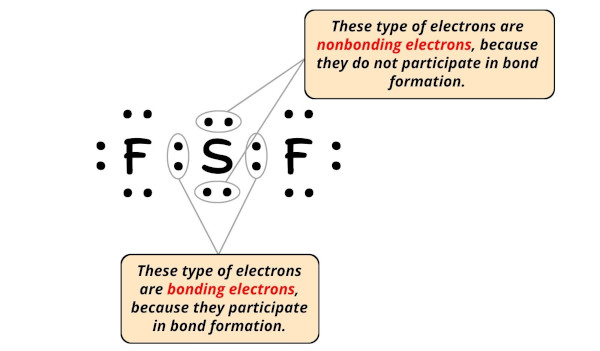
- For Sulfur:
Valence electron = 6 (as it is in group 16)
Nonbonding electrons = iv
Bonding electrons = 4 - For Fluorine:
Valence electron = 7 (as it is in group 17)
Nonbonding electrons = 6
Bonding electrons = 2
| Formal charge | = | Valence electrons | – | Nonbonding electrons | – | (Bonding electrons)/2 | ||
| S | = | 6 | – | 4 | – | 4/2 | = | 0 |
| F | = | 7 | – | 6 | – | 2/2 | = | 0 |
And then yous can come across above that the formal charges on sulfur too as fluorine are "zero".
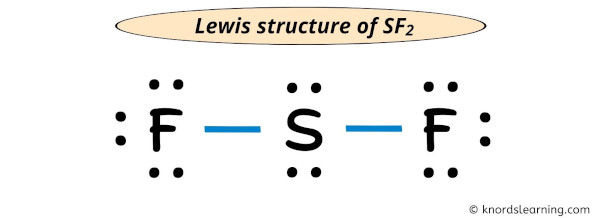
Hence, at that place volition not be any modify in the above structure and the in a higher place lewis structure of SF2 is the final stable structure only.
Each electron pair (:) in the lewis dot structure of SF2 represents the single bail ( | ). So the above lewis dot structure of SF2 can too be represented equally shown below.
Related lewis structures for your practise:
Lewis structure of IF5
Lewis structure of HNO3
Lewis structure of SCN-
Lewis structure of ClF3
Lewis structure of Cl2
Lewis Dot Structure For Sf2,
Source: https://knordslearning.com/lewis-structure-of-sf2/
Posted by: vincentcriagand59.blogspot.com


0 Response to "Lewis Dot Structure For Sf2"
Post a Comment